
The molecules of matter are composed of atoms, and their chemical bonds are constantly undergoing stretching and contracting vibrations. When light with a specific vibration frequency irradiates these molecules, if the vibration frequency of the light matches that of the chemical bonds in some of the molecules, the molecules will absorb the light, rendering it impermeable. By scanning with light of varying wavelengths, certain wavelengths will be absorbed, yielding a spectrum characteristic of the substance. This spectrum is called the light absorption spectrum. Just like human fingerprints, different substances have distinct absorption spectra. By measuring the absorption spectrum of an unknown substance, its composition and molecular structure can be determined.
An infrared spectrophotometer performs measurements based on this principle. Using infrared light with wavelengths ranging from 2.5 to 25 μm, it can analyze substances in any state, including solids, liquids, and gases.
Figure 1 shows the block diagram of an infrared spectrophotometer using the double-beam method. Light emitted by the light source is split by a semi-transparent mirror into a sample beam and a reference beam. The sample beam passes through the test sample; the reference beam is attenuated by an optical attenuator with uniform wavelength characteristics to match the intensity of the sample beam. The two beams are chopped at different frequencies by an optical chopper. The two beams of different frequencies are then recombined by another semi-transparent mirror and pass through a monochromator.
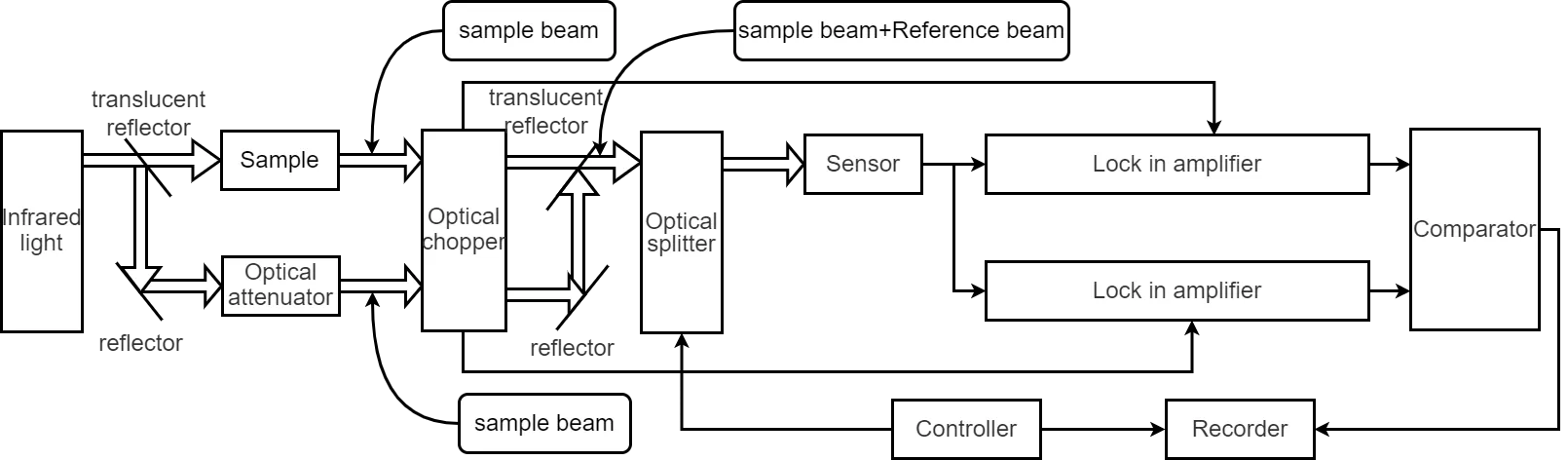
Fig. 1 Block diagram of an infrared spectrophotometer
The monochromator is equivalent to a band-pass filter (BPF) whose center frequency can be controlled externally. The controller issues commands to scan the wavelengths of light passing through it. The light from the monochromator is converted into an electrical signal by a sensor. The sample beam and reference beam are analyzed by two lock-in amplifiers using their respective chopping frequencies as reference signals. The comparator then calculates the ratio of the two beams. Finally, under the command of the controller, the light absorption spectrum of the sample is recorded by the recorder. The infrared spectrophotometer is a widely used analytical instrument, extensively applied in fields such as physical chemistry, medicine, biology, and environmental pollution monitoring.